 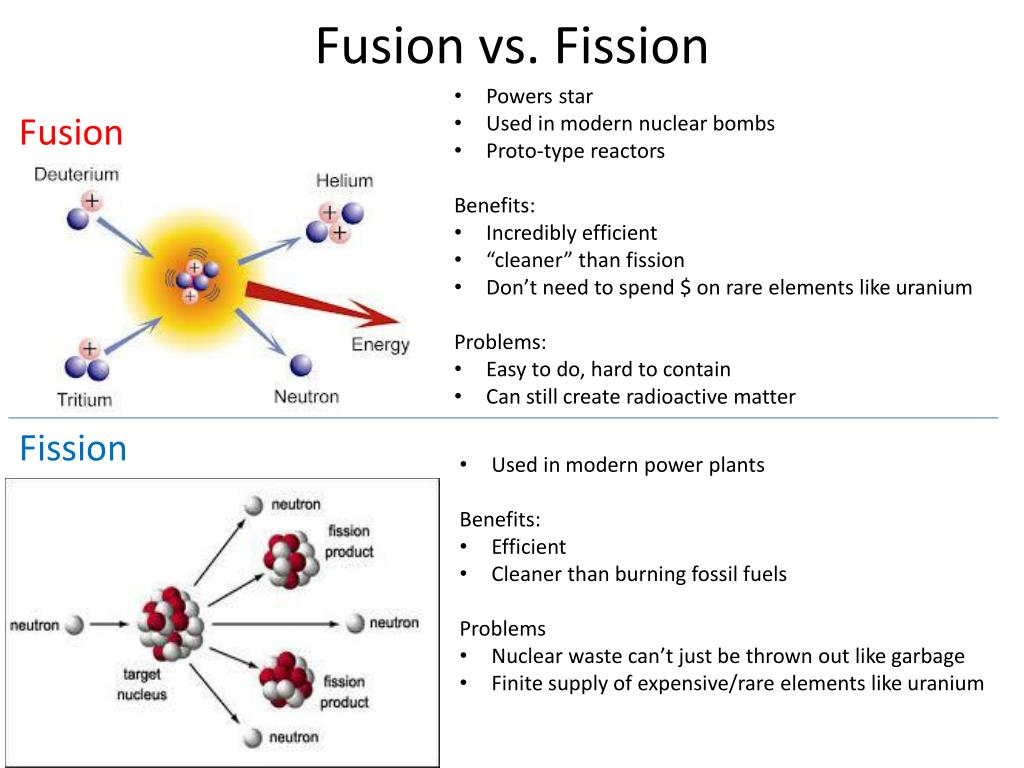
We predict that the overall process will give off five neutrons. (Because the atomic number of a neutron is zero, including five neutrons on the right does not change the overall sum of the subscripts.) Thus the balanced nuclear equation is as follows: Five neutrons should be products of the process for the mass numbers to balance. We need five more mass number units on the right. The superscripts on the left equal 240 (1 + 239) but equal 235 (204 + 31) on the right. The subscripts are already balanced: 0 + 94 = 94 and 79 + 15 = 94. In balanced nuclear equations, the sums of the subscripts on each side of the equation are the same, as are the sums of the superscripts. Using the data given, we can write the following initial equation: Write the balanced nuclear equation for the process and determine the number of neutrons given off as part of the reaction. Plutonium-239 can absorb a neutron and undergo a fission reaction to make an atom of gold-204 and an atom of phosphorus-31. 
(Artificial or induced radioactivity, in which neutrons are injected into a sample of matter that subsequently cause fission, was first demonstrated in 1934 by Irène Joliot-Curie and Frédéric Joliot, the daughter and son-in-law of Marie Curie.) Thus by the careful addition of extra neutrons into a sample of uranium, we can control the fission process and obtain energy that can be used for other purposes. The overall nuclear equation, with energy included as a product, is then as follows: The reaction can be controlled because the fission of uranium-235 (and a few other isotopes, such as plutonium-239) can be artificially initiated by injecting a neutron into a uranium nucleus. Nuclear energy involves the controlled harvesting of energy from fission reactions. 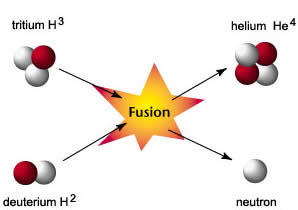
If this energy could be properly harvested, it would be a significant source of energy for our society. Nuclear reactions give off billions of kilojoules per mole. Compare it to combustion reactions of hydrocarbons, which give off about 650 kJ/mol of energy for every CH 2 unit in the hydrocarbon - on the order of hundreds of kilojoules per mole. This is an extraordinary amount of energy. (For the units to work out, mass must be expressed in units of kilograms.) That is, 16.5 billion kJ of energy is given off every time 1 mol of uranium-235 undergoes this nuclear reaction. In the course of the chemical reaction for uranium, the mass difference is converted to energy, which is given off by the reaction: Where c is the speed of light, or 3.00 × 10 8 m/s. Where did this mass go?Īccording to Albert Einstein’s theory of relativity, energy ( E) and mass ( m) are related by the following equation: If we compare the mass of the reactant (235.0439) to the masses of the products (sum = 234.8605), we notice a mass difference of −0.1834 g, or −0.0001834 kg. Consider the following nuclear equation, in which the molar mass of each species is indicated to four decimal places: Where does this energy come from? If we could precisely measure the masses of the reactants and products of a nuclear reaction, we would notice that the amount of mass drops slightly in the conversion from reactants to products. Nuclear changes occur with a simultaneous release of energy.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
